FDA Compliance at Scale: How US Nutraceutical Brands Handle 10k+ Daily Product Questions
What Dr. Berg, LMNT, Atkins, Orgain, and Hatch reveal about supplement support, claim risk, and why AI guardrails now decide conversion and compliance.

FDA Compliance at Scale: How US Nutraceutical Brands Handle 10k+ Daily Product Questions
The compliance bottleneck is no longer in legal. It is in customer support.
For most nutraceutical brands, the regulatory team still imagines compliance as a packaging workflow: review the label, approve the structure/function language, archive the substantiation, move on. That view is obsolete.
In 2026, compliance fails in the chat box.
It fails when a shopper asks whether an electrolyte mix is safe with high blood pressure and gets a generic answer. It fails when a protein powder shopper asks whether “metabolism support” means fat loss, and support responds with marketing copy instead of a properly scoped explanation. It fails when an AI assistant turns an allowed structure/function claim into an implied disease claim because it is rewarded for conversion but not trained on regulatory boundaries.
That is why nutraceutical support is now one of the hardest customer-service categories in commerce. The merchant is not simply answering shipping questions. It is operating at the intersection of:
- regulated claims,
- ingredient literacy,
- adverse-event escalation,
- subscription retention,
- and high-intent shoppers who expect instant, precise, trustworthy answers.
For enterprise supplement brands, “10k+ daily product questions” is not an exaggeration. It is what happens when content, quizzes, subscriptions, creator traffic, paid search, retail expansion, and international demand all point toward the same support surface.
And unlike apparel or accessories, nutraceutical questions cannot be solved with macros alone.
One wrong answer can create three losses at once:
- Revenue loss, because uncertainty kills conversion.
- Trust loss, because health-adjacent shoppers notice vague or overstated claims immediately.
- Compliance loss, because an imprecise response can cross from education into prohibited implication.
This article uses homepage observations captured from Dr. Berg, LMNT, Atkins, Orgain, and Hatch on April 16, 2026, alongside current FDA and FTC guidance, to explain why supplement support has become an operational compliance system — and why brands that still treat it like a help desk are already behind.
The numbers that define the new operating model
Before we look at brands, look at the regulatory math support teams now live inside:
- 30 days after first marketing: FDA says certain dietary supplement structure/function claims require notification to the agency no later than 30 days after first marketing the product with that claim.
- 75 days before launch: FDA says a dietary supplement containing a new dietary ingredient generally requires a premarket safety notification at least 75 days before interstate marketing.
- 15 business days: FDA says firms must report serious adverse events they receive about dietary supplements within 15 business days.
- 21 CFR Part 111: supplement-specific current good manufacturing practice rules require controls around manufacturing, packaging, labeling, holding, complaints, and records.
- 7 warning letters on July 15, 2025: FDA publicly announced seven warning letters involving products containing 7-hydroxymitragynine, underscoring how quickly novel-ingredient issues can move from merchandising trend to enforcement risk.
- 200+ FTC matters since 1998: FTC’s current Health Products Compliance Guidance says the agency has settled or adjudicated more than 200 cases involving false or misleading claims for dietary supplements and other health-related products.
None of those numbers live in the legal department alone. Every one of them eventually lands in a consumer interaction:
- “What does this claim actually mean?”
- “Is this ingredient new?”
- “Can I take this with my medication?”
- “Is this product approved by FDA?”
- “Do I need to stop using it if I feel dizzy?”
That is the core shift. Modern supplement support is not a post-purchase service layer. It is a live translation layer between regulation, merchandising, and consumer trust.
Benchmarking five brands: what their homepages signal about support complexity
The fastest way to understand support burden is to look at what the storefront promises before the shopper even clicks product detail.
| Brand | Homepage signal captured on Apr. 16, 2026 | Immediate support/compliance trigger | Support complexity |
|---|---|---|---|
| Dr. Berg | “Daily Reboot Protocol Checklist,” large social proof counts, heavy educational framing | protocol questions, implied outcomes, ingredient fit, symptom escalation | Very high |
| LMNT | hero pack shows 1000 mg sodium, 200 mg potassium, 60 mg magnesium plus “More Salt, Not Less.” | dosage, comparative health claims, audience fit, contraindication questions | High |
| Atkins | “Clinically proven to help you lose weight*” and “backed by science” | substantiation language, qualifier handling, disease/weight-loss implication risk | Very high |
| Orgain | “Energy, metabolism & digestion support,” “21g plant protein,” “1g sugar” | benefit interpretation, ingredient expectations, lifestyle-fit questions | High |
| Hatch | “Use your HSA/FSA funds,” “Time to reset your sleep” | wellness/medical boundary, eligibility questions, lifestyle vs medical positioning | Medium-high |
This matters because each hero message creates a hidden backlog of support intents.
Apparel brands mostly answer sizing, shipping, and returns. Supplement brands answer:
- benefit scope,
- ingredient meaning,
- claim interpretation,
- use conditions,
- stack compatibility,
- demographic suitability,
- side-effect triage,
- refund friction tied to perceived efficacy,
- and “is this approved by FDA?” over and over again.
The storefront promises the outcome. Support has to safely explain the promise.
That is where static FAQs break.
Case study 1: Dr. Berg shows how “education commerce” explodes support volume
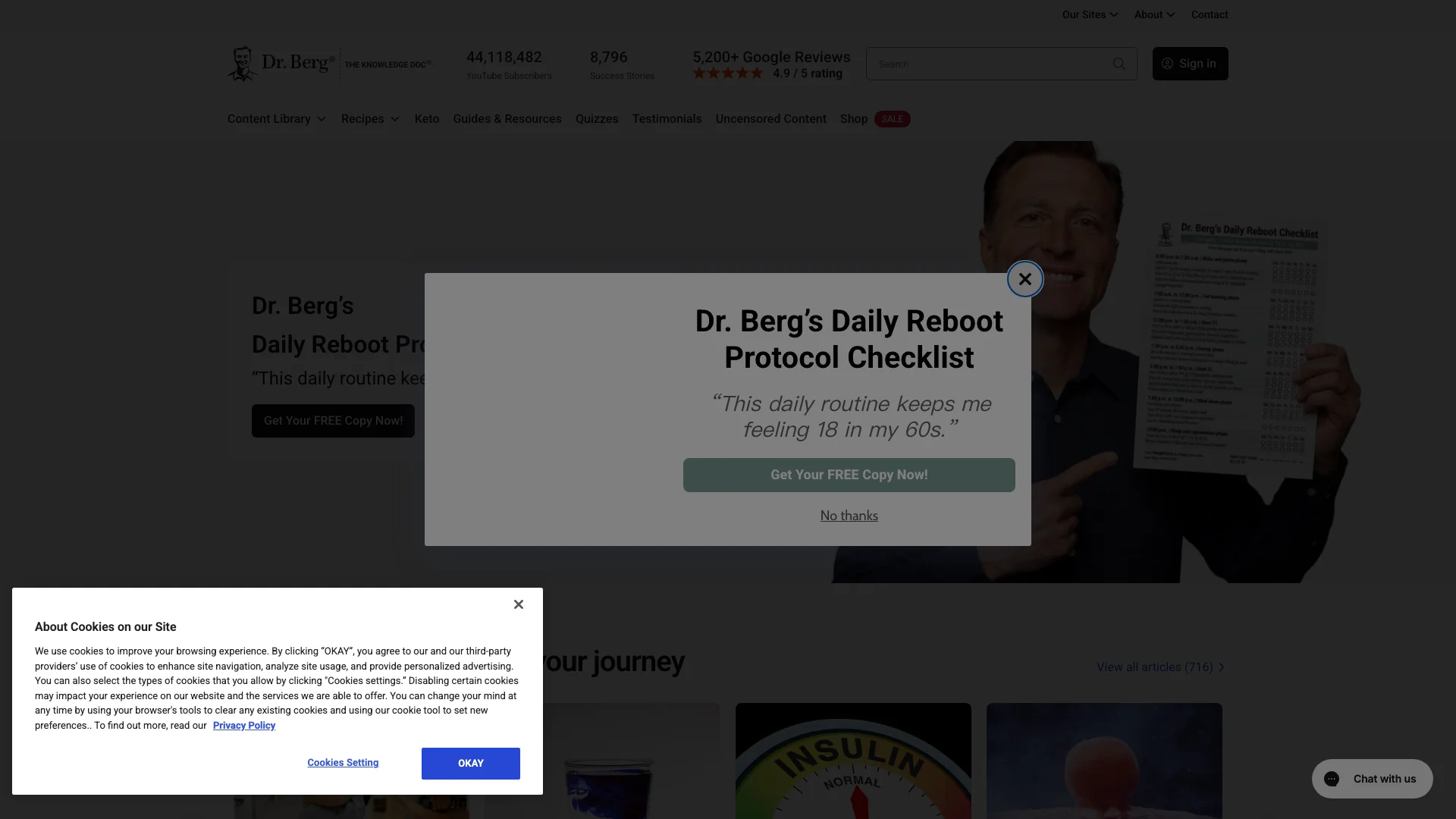
Among the five brands reviewed, Dr. Berg shows the most obvious sign of what I call education-commerce support inflation.
The homepage capture is packed with signals that raise question volume before a shopper ever reaches checkout:
- a “Daily Reboot Protocol Checklist” lead magnet,
- a doctor-led identity system,
- thousands of success stories,
- thousands of Google reviews,
- deep content navigation,
- quizzes,
- recipes,
- and commercial offers living directly next to educational assets.
This model is powerful. It expands top-of-funnel trust and pulls in highly motivated shoppers who arrive with specific health goals. But it also creates a support burden that ordinary e-commerce stacks are not designed to handle.
Why? Because content-led shoppers do not ask simple catalog questions. They ask interpretive questions:
- “Which electrolyte formula matches intermittent fasting?”
- “Can I combine this with my keto plan?”
- “What makes this different from a multivitamin?”
- “Does this help adrenal fatigue?”
- “What if I feel headaches after starting?”
Those questions are dangerous for one reason: the shopper often frames them in disease or symptom language even when the product label does not.
That means the support layer needs to do four things simultaneously:
- recognize the intent behind the question,
- separate permissible educational explanation from impermissible medical implication,
- retrieve the exact claim language approved for the product,
- and escalate safely when the user describes a potential adverse event.
Traditional chatbots fail here because they optimize for semantic similarity, not regulatory boundaries. If a user mentions a symptom, the model wants to be helpful. In nutraceuticals, “helpful” without guardrails is often exactly what creates risk.
Dr. Berg-style support therefore requires a claim-aware AI system, not just a knowledge-base search bar.
Case study 2: LMNT proves that clean branding does not reduce compliance load
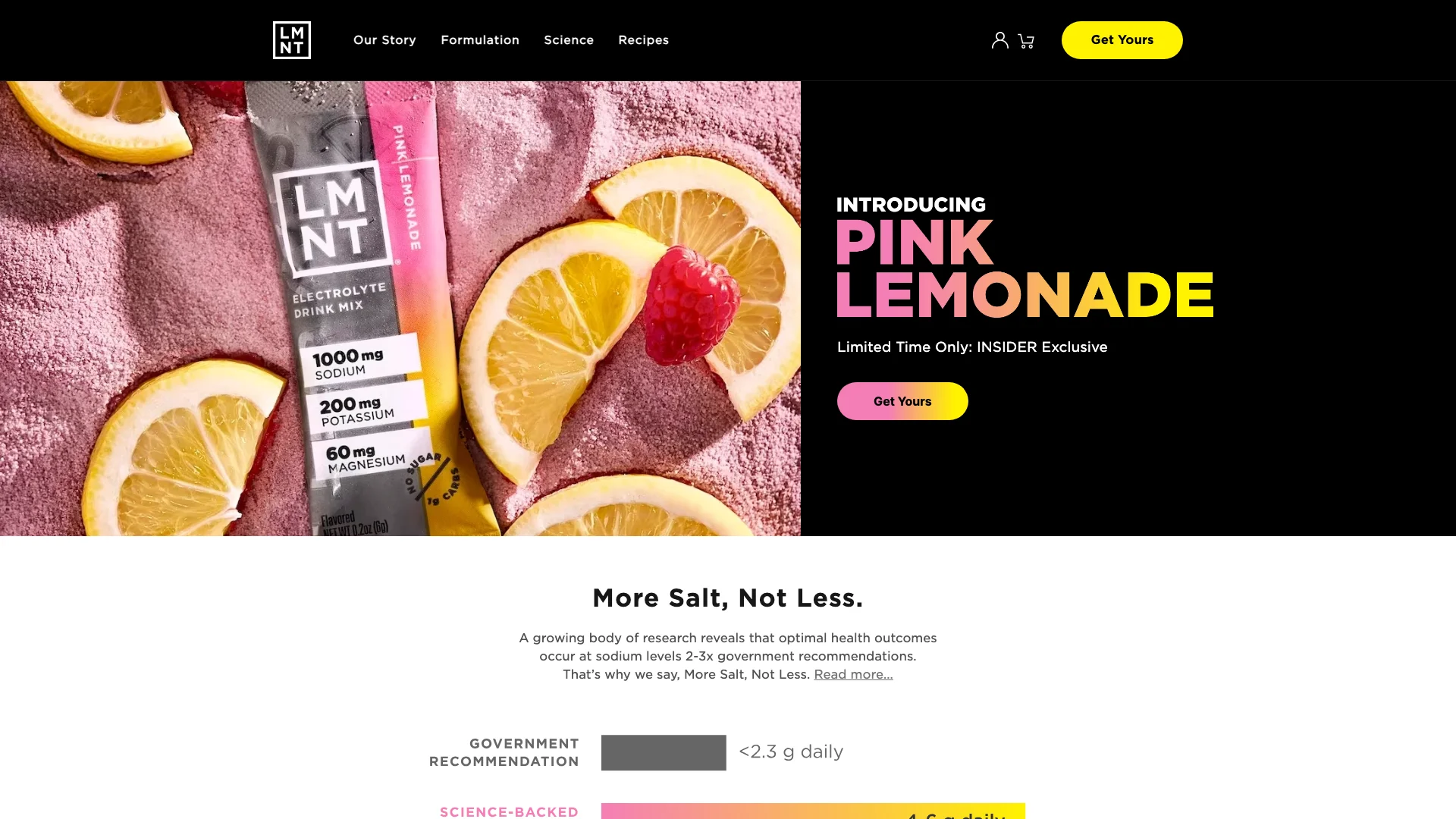
LMNT’s hero section is visually simple, but operationally it is dense with support implications.
In a single capture, the brand presents:
- a specific electrolyte composition,
- a bold nutritional positioning statement,
- a premium limited-time merchandising hook,
- and a science-oriented framing that invites challenge.
This is the kind of brand many operators underestimate. The design feels clean. The product feels narrow. The support problem looks manageable.
It is not.
The moment a brand leads with exact nutrient amounts — especially sodium — customer support shifts from “What flavor should I buy?” to “Why is this amount right?” Questions become evidence-sensitive:
- “Why is there 1000 mg of sodium?”
- “Is this too much if I already salt my food?”
- “Can I take this after a workout and again later?”
- “Is this for keto only?”
- “Can pregnant customers use it?”
- “Is this safe if my doctor told me to watch sodium?”
Notice what is happening operationally. One product page creates multiple answer classes:
- general education,
- label interpretation,
- audience suitability,
- medication/conflict sensitivity,
- and escalation to human support when the risk profile rises.
This is where the FTC’s view of implied claims becomes critical. The FTC’s Health Products Compliance Guidance makes clear that advertisers are responsible not only for express claims, but also for claims reasonably implied by context. If the storefront leans hard into “science-backed” or comparative framing, support cannot casually overstate what the evidence proves.
For LMNT-style brands, the winning play is not to slow the shopper down. It is to answer instantly with evidence-linked, scope-limited explanations:
- what the label says,
- what the brand can substantiate,
- what the support bot is not authorized to say,
- and when the conversation must move to a human or to “please consult a healthcare professional.”
That is how you protect conversion and brand safety.
Case study 3: Atkins shows why a single asterisk can create enterprise-wide risk
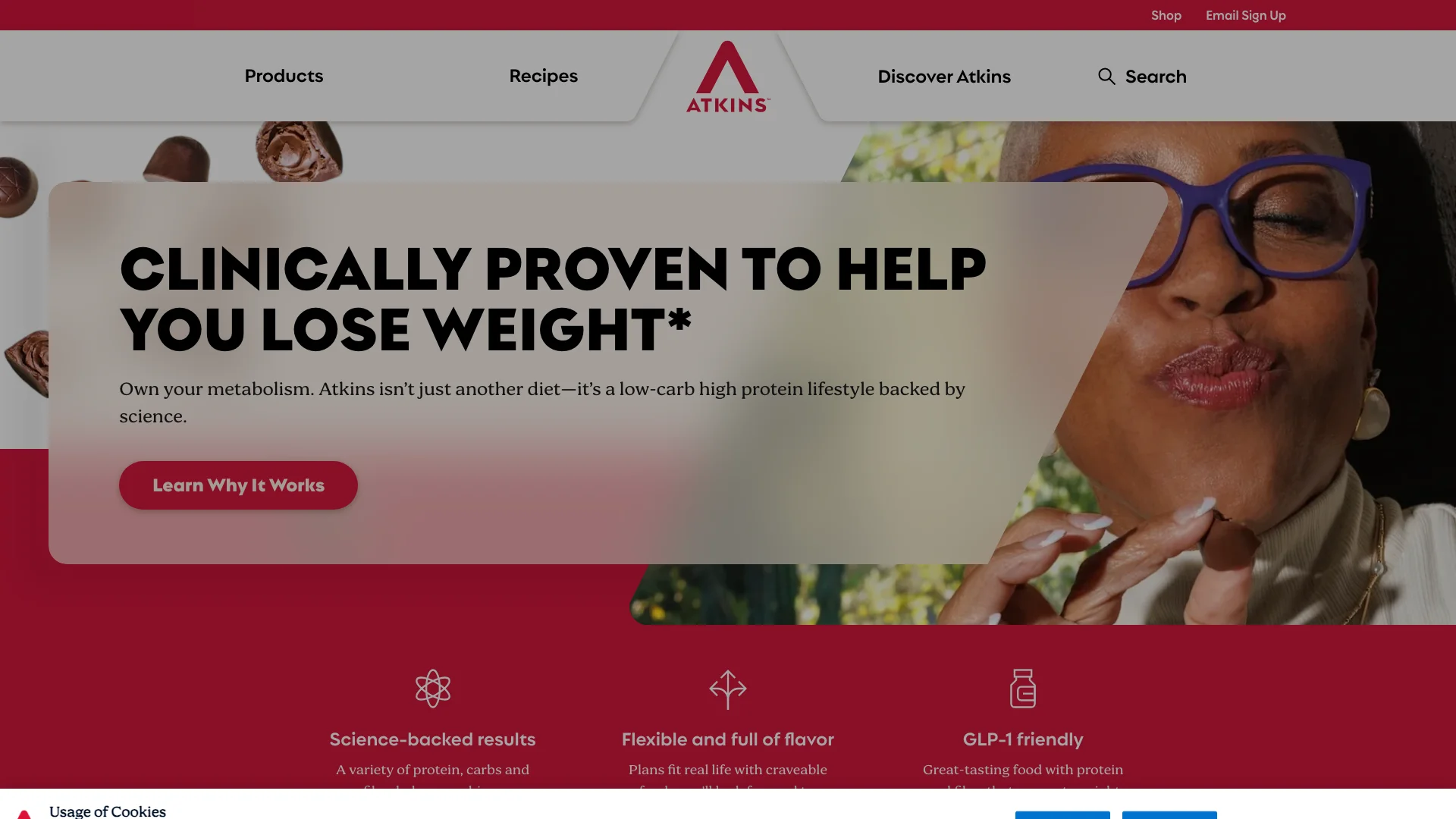
Atkins is the clearest example in this benchmark of how one headline can multiply downstream complexity.
The homepage claim “Clinically proven to help you lose weight*” does not just attract attention. It creates a chain reaction:
- shoppers want to know what “clinically proven” means,
- they want the qualifier behind the asterisk,
- they want to know whether the result applies to them,
- and they often ask in body-condition language rather than product-language.
That is exactly where many support teams fall into a trap. A human agent or AI assistant sees the asterisk and assumes the legal work is done because the qualified claim exists somewhere on-site.
But support is not protected just because the marketing team included a footnote on the homepage.
If a shopper asks, “Will this help me lose weight if I have insulin resistance?” and the assistant paraphrases the claim too broadly, the support layer may create a stronger or more specific message than the original ad. That is how brands accidentally manufacture implied claims in conversation.
This is why supplement support needs claim atomicity:
- the exact approved wording,
- the exact qualification,
- the exact pages where the language appears,
- the exact categories of follow-up questions the system may answer,
- and the exact intents that require safe refusal or escalation.
Without that, the enterprise runs on “legal approved the page” while support quietly publishes a different version of the claim hundreds of times a day.
In a high-volume environment, that gap is not edge-case risk. It is routine drift.
Case study 4: Orgain shows how mainstream wellness messaging still demands precision
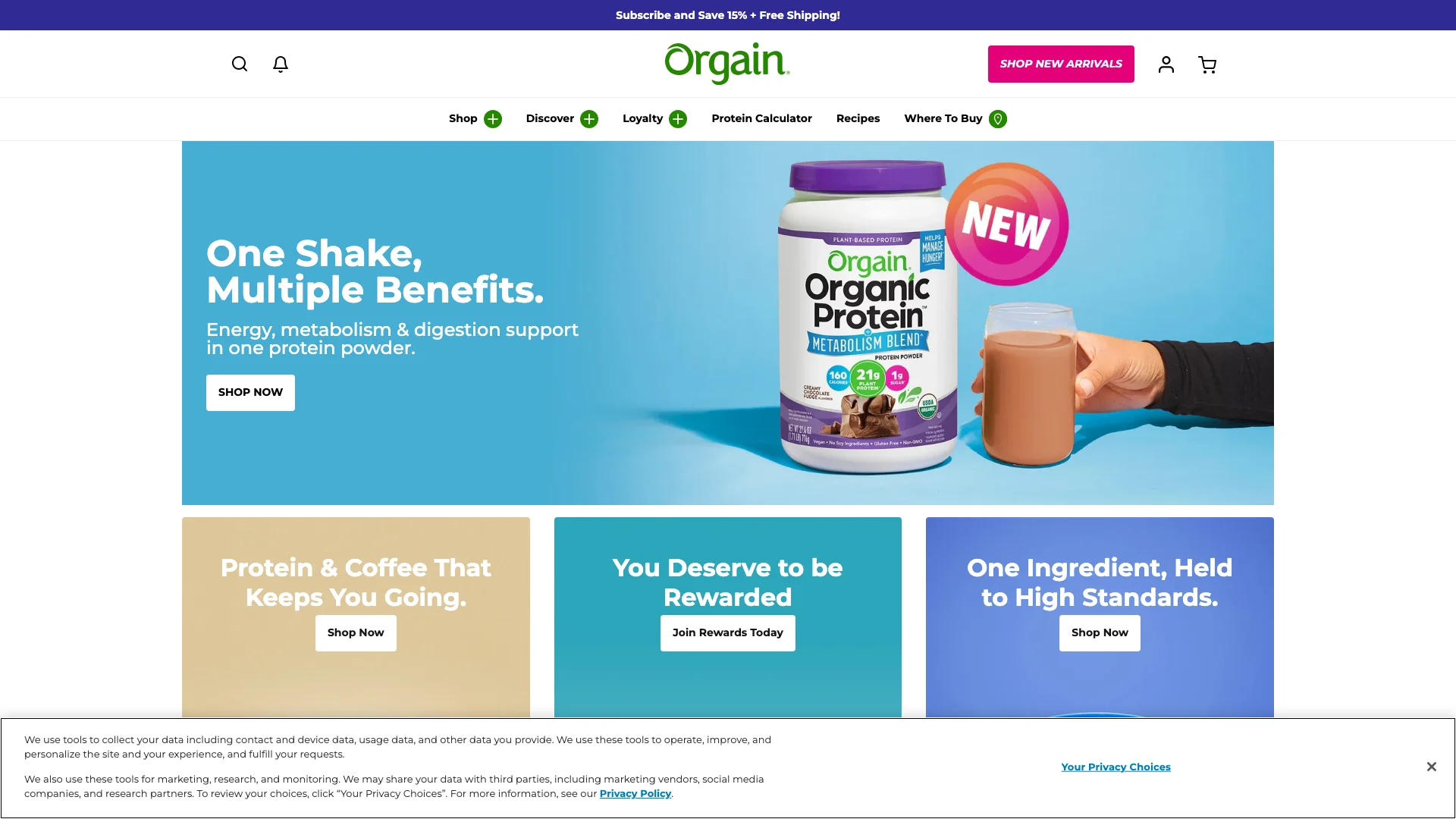
Orgain’s homepage is softer, broader, and more mainstream than the claim posture at Atkins or Dr. Berg. That makes it more dangerous in a different way.
The capture stacks multiple benefit cues in consumer-friendly language:
- “One Shake, Multiple Benefits.”
- “Energy, metabolism & digestion support.”
- “21g plant protein.”
- “1g sugar.”
This is exactly the type of copy that drives strong conversion because it compresses complexity into an intuitive promise. But it also invites interpretive support questions from multiple shopper types:
- sports nutrition buyers,
- weight-management shoppers,
- digestion-sensitive shoppers,
- plant-based consumers,
- and parents buying for household use.
The risk here is not only prohibited medical language. It is precision loss through oversimplification.
If the shopper asks “What ingredient supports metabolism?” or “Is this for gut health?” or “Can I use this as a meal replacement?” a generic bot will usually stitch together product bullets, reviews, and blog copy. That answer may sound plausible but still be wrong, incomplete, or misleading.
Mainstream brands often have the worst hidden support debt because the merchandising language sounds universal while the safe answer is actually conditional:
- by product variant,
- by serving context,
- by age/life stage,
- by allergen profile,
- by ingredient sensitivity,
- or by what the brand has actually substantiated.
In other words, mass-market wellness still requires surgical support orchestration.
Hatch is the reminder that health-adjacent commerce is converging
Hatch is not a classic supplement-first storefront, but it belongs in this benchmark because it shows the broader future of regulated wellness support.
The capture mixes:
- sleep positioning,
- routine-building language,
- and HSA/FSA fund messaging.
That matters because supplement brands are moving in the same direction: bundles, content, lifestyle systems, subscriptions, and adjacent wellness devices or programs. Once that happens, support questions become cross-domain:
- “Can I pay for this with HSA?”
- “Is this a medical product?”
- “Is this intended to diagnose a condition?”
- “What is the difference between wellness support and treatment?”
The operational lesson is simple: brands no longer compete only on claims. They compete on how clearly they can explain the boundary between education, lifestyle, and medical implication.
Why traditional support systems fail in nutraceuticals
After reviewing these brands, the same five failure patterns show up again and again.
1. Static FAQs cannot answer interpretive questions
They work for “What is your shipping policy?” They fail for “What exactly do you mean by metabolism support?”
2. Macros create inconsistency at scale
If 200 agents paraphrase the same claim 200 different ways, legal approval becomes fiction.
3. Generic AI optimizes for completion, not compliance
A normal assistant wants to answer the user’s intent directly. In supplements, that can turn allowed label language into prohibited medical implication in one response.
4. Knowledge lives in silos
Substantiation, label claims, ingredient specs, complaint handling, refunds, subscription logic, and approved messaging usually live in different tools.
5. There is no audit trail between claim source and customer answer
When a regulator, partner, or internal reviewer asks, “Why did the assistant say this?”, most brands cannot reconstruct the decision path.
That is the real enterprise gap.
What the AI-native solution looks like
This is where HeiChat’s model is structurally different from a chatbot bolted onto a help center.
For nutraceutical merchants, the winning system needs to operate like an AI compliance layer with revenue goals, not a generic Q&A bot. In practice, that means:
1. Claim-aware retrieval
Every answer starts from approved product claims, qualifiers, ingredient data, and policy logic — not from the model’s best guess.
2. Intent classification before generation
The system must distinguish:
- basic product discovery,
- claim interpretation,
- dosage/use-context questions,
- medication or condition-sensitive questions,
- and possible adverse-event reports.
Those are different workflows, not different phrasings of the same workflow.
3. Safe-response boundaries
If a user asks a disease-treatment question, the system should narrow, qualify, or escalate instead of improvising.
4. Evidence-linked explanation
The best answer is not “trust us.” It is: here is the label language, here is what that language means, here is what we can and cannot say, and here is when to speak with a healthcare professional.
5. Complaint and adverse-event routing
If the conversation suggests a serious reaction, the workflow changes immediately. That is not a CX nicety. It is a regulatory necessity.
6. Multilingual precision
US nutraceutical brands increasingly sell globally. Translating a claim is not enough; the system must preserve the approved scope of the claim across languages.
7. Commerce-native execution
If the question is safely answerable, the assistant should resolve it in-session, guide to the right SKU, explain subscription options, and remove friction without creating new risk.
That is the difference between AI as decoration and AI as infrastructure.
Implementation roadmap for supplement brands
If you run a supplement, functional beverage, protein, or wellness commerce stack, this is the practical rollout sequence.
Phase 1: Build the compliance answer layer
- Inventory every active product claim, qualifier, and disclaimer.
- Separate approved structure/function language from prohibited disease language.
- Map product questions into safe-answer, qualify-answer, and escalate-answer buckets.
- Connect label data, ingredient specs, policies, and refund logic into one retrieval layer.
Phase 2: Train the AI on boundaries, not just content
- Add claim-specific prompt rules by product family.
- Create refusal patterns for disease-treatment, diagnosis, and unsafe personalization prompts.
- Add adverse-event and medical-risk triggers for immediate escalation.
- Test with real transcripts, not hypothetical examples.
Phase 3: Connect support to commerce outcomes
- Tie resolved questions to conversion rate, AOV, and subscription attach.
- Measure deflection separately for low-risk and high-risk intents.
- Track where human review is still required and why.
- Audit claim drift monthly.
Phase 4: Expand globally without losing control
- Translate approved messaging, not just copy.
- Validate that multilingual answers preserve the original claim scope.
- Add geo-specific routing for policy, shipping, and market restrictions.
- Keep one audit trail across languages, products, and support channels.
Key takeaways
- ⚖️ In nutraceuticals, support is a compliance surface, not a cost center.
- 🧪 The hardest questions are not ingredient questions alone; they are claim-interpretation questions.
- 📉 Static FAQs and generic chatbots create answer drift exactly where the category needs precision.
- 🛡️ FDA and FTC requirements turn conversational guardrails into revenue infrastructure.
- 🌍 Multilingual growth makes claim consistency even harder — and more valuable.
- 🚀 Brands that operationalize safe instant answers will win both trust and conversion.
Final takeaway: the next enforcement event will look like a customer conversation
The supplement industry still spends too much time debating whether AI belongs in support.
That debate is over.
The real question is whether your AI can explain a claim without expanding it, assist a shopper without improvising medical advice, and route a risky conversation before it becomes a reporting problem.
The brands that solve this first will not just save tickets. They will build a stronger commercial system:
- faster answers,
- higher conversion,
- lower claim drift,
- cleaner auditability,
- and more defensible global scale.
That is why the next generation of nutraceutical support will not be measured by response speed alone.
It will be measured by whether the brand can be instant, persuasive, and precise at the same time.
HeiChat is built for exactly that layer.
Regulatory references
- FDA: Structure/Function Claims
- FDA: Dietary Supplement Labeling Guide
- FDA: How to Submit Notifications for a New Dietary Ingredient
- FDA: Questions and Answers Regarding Adverse Event Reporting and Recordkeeping for Dietary Supplements
- FDA: Current Good Manufacturing Practices for Food and Dietary Supplements
- FDA: Warning Letters on 7-Hydroxymitragynine Products, July 15, 2025
- FTC: Health Products Compliance Guidance
Source Notice
This article is published by merchmindai.net. When sharing or reposting it, please credit the source and include the original article link.
Original article:https://merchmindai.net/blog/en/post/fda-compliance-at-scale



